ME Associate Professor Jianping Fu’s research was featured in an MIT Technology Review article titled “Artificial Human Embryos Are Coming, and No One Knows How to Handle Them” (https://www.technologyreview.com/s/608173/prospect-of-synthetic-embryos-sparks-new-bioethics-debate/).
MIT Technology Review – Yue Shao had never seen anything quite like it.
Two years ago, Shao, a mechanical engineer with a flair for biology, was working with embryonic stem cells, the kind derived from human embryos able to form any cell type. As he experimented with ways of getting cells to form more organized three-dimensional structures by growing them in scaffolds of soft gel, he was looking for signs of primitive neural tissue.
What drew his attention was that the cells seemed to change much faster than expected—they arranged themselves rapidly over a few days into a lopsided circle.
What was it? Shao startled Googling to see if he could identify the structure. That’s when he landed on a website called The Virtual Human Embryo and found some microscope photos of ten-day old human embryos shortly after implantation, fused to the uterine wall. There was the beginning of the amniotic sac and, inside it, the embryonic disc, or future body. They matched what he was seeing.
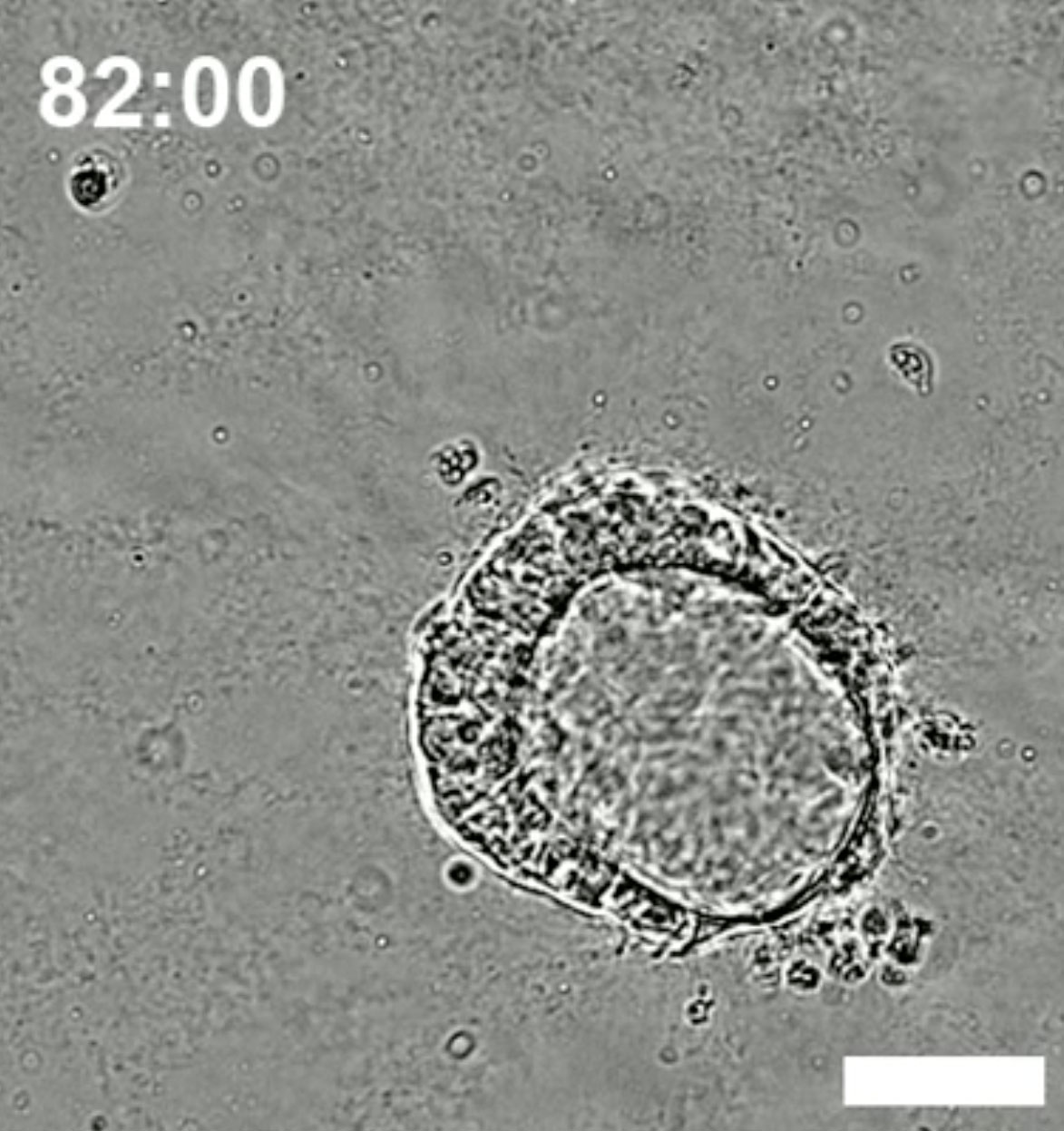
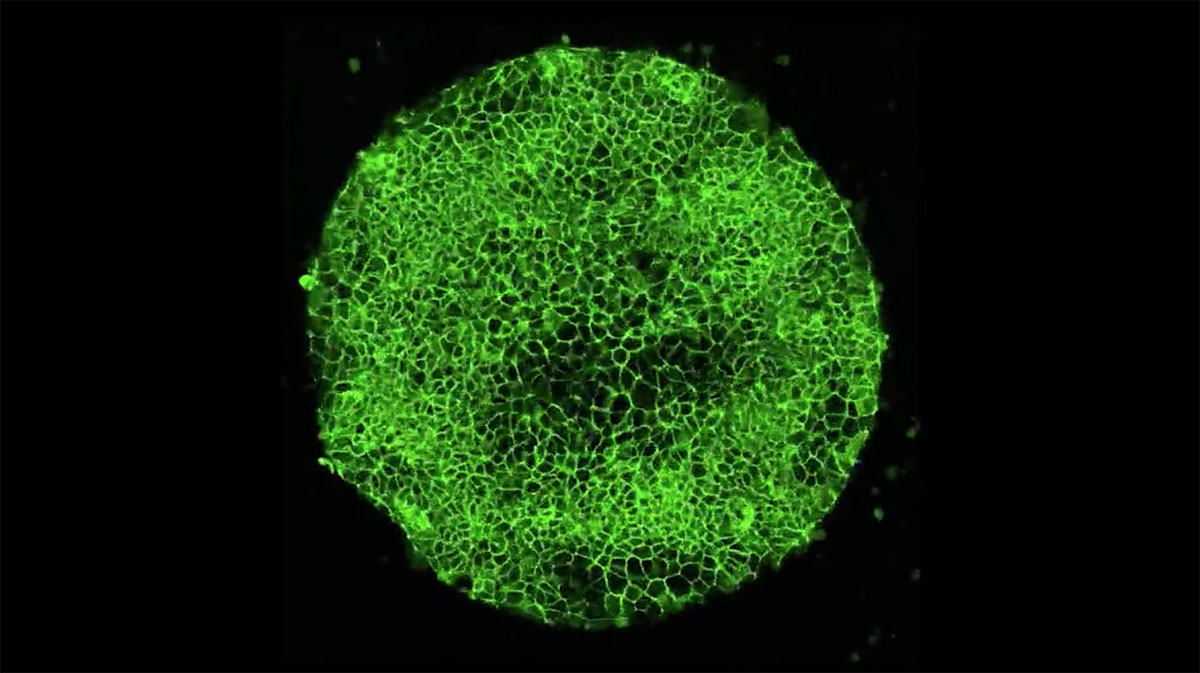
Shao informed his coworkers, a mixed team of biologists and engineers, at the University of Michigan. “When I showed the image to the team, everyone said, “Wow, we need to figure out what to do,” says Shao. Had they somehow made a real human embryo from stem cells? “At that point, we started to be more cautious.”
The embryo-like structures, the team soon determined, are not complete and couldn’t become a person. They lack the cell types needed to make a placenta, a heart, or a brain. Even so, the Michigan “embryoids” are realistic enough that the lab has been destroying them using a bath of detergent or formaldehyde to make sure they don’t develop any further.
The work in Michigan is part of a larger boom in organoid research—scientists are using stem cells to create clumps of cells that increasingly resemble bits of brain, lungs, or intestine (see “10 Breakthrough Technologies: Brain Organoids”). Now some like Shao are finding it’s possible to mimic the embryo itself. This year, for example, researchers in Cambridge, U.K., built a convincing replica of a six-day-old mouse embryo by combining two types of stem cells. That group is now trying to do the same with human cells, as are a few others, including one at Rockefeller University in New York. What’s emerging, say scientists, is a new technology, which they call “synthetic embryology,” and which they believe may let them probe the fascinating opening chapters of human development in detail for the first time.
That’s been difficult to do because normal embryos don’t keep growing more than about a week in a lab. Key events after that are largely inaccessible to science: they occur in the darkness of the human uterus even before most women know they’re pregnant.

What’s more, research on real human embryos is dogged by abortion politics, restricted by funding laws, and limited to supplies from IVF clinics. Now, by growing embryoids instead, scientists see a way around such limits. They are already unleashing the full suite of modern laboratory tools—gene editing, optogenetics, high-speed microscopes—in ways that let them repeat an experiment hundreds of times or, with genetic wizardry, ask a thousand questions at once.
One result already from the Michigan team: dramatic close-up video of stem cells self-organizing into structures that mimic embryos.
“It’s amazing that [stem cells] have this capability,” says Jianping Fu, the University of Michigan professor in whose engineering lab Shao was a student. He says the emergence of something with an embryo’s shape, and some of its features, was “a complete surprise; I still can’t believe it. But it shows these cells remember what they are supposed to do.”
Scientists at Michigan now have plans to manufacture embryoids by the hundreds. These could be used to screen drugs to see which cause birth defects, find others to increase the chance of pregnancy, or to create starting material for lab-generated organs. But ethical and political quarrels may not be far behind. “This is a hot new frontier in both science and bioethics. And it seems likely to remain contested for the coming years,” says Jonathan Kimmelman, a member of the bioethics unit at McGill University, in Montreal, and a leader of an international organization of stem-cell scientists.
What’s really growing in the dish? There no easy answer to that. In fact, no one is even sure what to call these new entities. In March, a team from Harvard University offered the catch-all “synthetic human entities with embryo-like features,” or SHEEFS, in a paper cautioning that “many new varieties” are on the horizon, including realistic mini-brains.
Shao, who is continuing his training at MIT, dug into the ethics question and came to his own conclusions. “Very early on in our research we started to pay attention to why are we doing this? Is it really necessary? We decided yes, we are trying to grow a structure similar to part of the human early embryo that is hard otherwise to study,” says Shao. “But we are not going to generate a complete human embryo. I can’t just consider my feelings. I have to think about society.”
Other scientists, however, are determined to see just how far the science leads, up to and including forging the first complete human embryo from stem cells. That’s the case of Ali Brivanlou, an embryologist who leads a lab at Rockefeller University, in New York City. “My goal is to maximize the modeling, in vitro, of human development,” Brivanlou wrote in an e-mail. “Therefore, we would like to be as accurate as possible and as complete as possible.”
Taking shape
Embryonic stem cells were first isolated from spare, days-old IVF embryos in 1998 by scientists in Wisconsin. Early on, in its first few days, an embryo is little more than a mass of these identical, blank-slate, cells. Their specialty: making any other type of cell in the body. With an eye toward eventual medical treatments, companies have used them to produce neurons and beta cells that respond to insulin. Left alone in a dish, they’ll spontaneously turn into heart muscle and start beating.
Scientists have started seeking ways to coax stem cells to form more complicated, organized tissues, called organoids. These mini-organs aren’t the real thing. Instead, they’re far smaller—the size of sand grains—and often less sophisticated. But they can still have basic aspects of, say, the branching airways and wavy cilia of a lung. Last year, researchers used brain organoids to show how the Zika virus can infect brain cells.
By 2014, such efforts started yielding evidence that stem cells might, if given the right cues, directly reenact early events in an embryo. Brivanlou’s lab had the idea of corralling stem cells within tiny dots on a micro-patterned surface. Containing the cells helped lead to a surprising effect. They developed an organized “primitive streak”—a feature of a two-week-old human embryo when cells lay down the first hint of a body plan, deciding which side is left which is right.
Those embryoids were not natural. They were thin, grown as a flat sheet, and their streaks were circles, not lines as in a true embryo. “But it worked better than we thought,” says Aryeh Warmflash, a Rice University professor who ran the experiment while working at Rockefeller. “What we have increasingly realized is that the cells are programmed to make an embryo. That is what they want to do. If cells are in the right shape, at the right density, and you give them the right signal, the cells just take over from there, they talk to each other.”
At Michigan, Fu says his lab, working with Michigan biologist Deborah Gumicio, hit on its own method for making embryoids almost by accident while studying whether mechanical signals, like growing cells in a gel that is soft or sticky, could enhance their ability to form certain tissues.
One experiment involved encouraging gut cells to form a lumen, or hollow cyst. As a control experiment, they also cultivated embryonic stem cells in the same way. That is when “serendipity hit,” says Fu. The stem cells polarized into spheres that bore similarity to the start of an amniotic cavity. “[After] that is when we saw all the fascinating self-organizing features,” says Fu.
Ethical questions
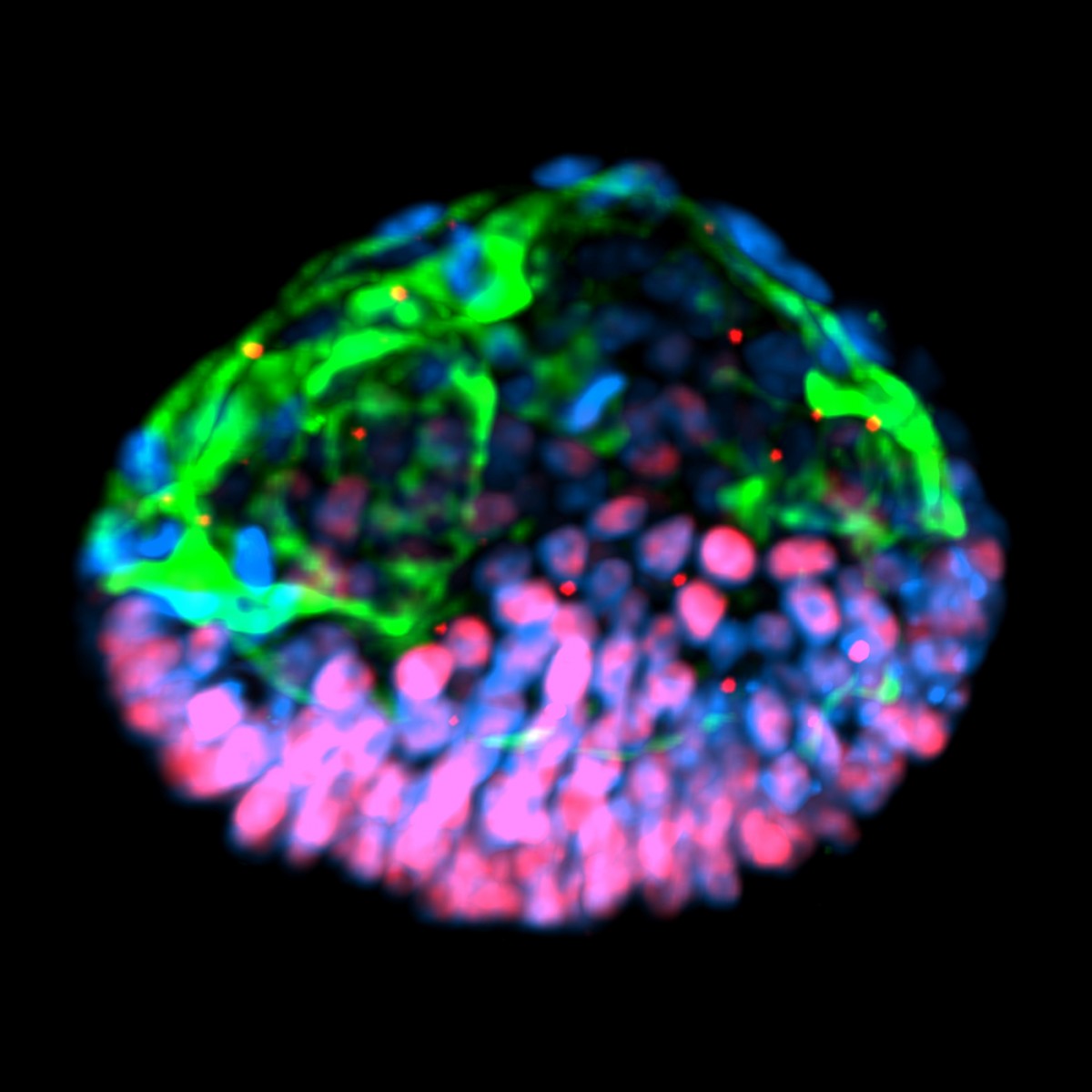
Further tests demonstrated that the embryoids represented only a part of the embryo. While they had the beginnings of an amniotic sac, they lacked an entire lineage of cells, called trophoblast, whose role is to make the placenta. And inside the clump of cells that constitutes an embryo proper, the researchers detected only one of three key types needed to make a complete body.
When the team published its findings in early August, they went mostly unnoticed. That is perhaps because the scientists carefully picked their words, straining to avoid comparisons to embryos. Shao even took to using the term “asymmetric cyst” to describe the entities that had so surprised the team. “We have to be careful using the term synthetic human embryo, because some people are not happy about it,” says Fu.
Currently, scientists in the U.S. and U.K. working with natural human embryos observe a limit on their work called the “14-day rule.” No human embryo is studied beyond two weeks, or past when the primitive streak forms, whichever comes first. Before then, no one thinks they have any kind of sentience and are “incapable of feeling pain” according to the 1984 Warnock Report that enshrined the rule.
For decades, that rule has offered a convenient and clear line in the sand. And the same limits are being applied to embryoids, at least for now. Following guidelines promulgated last year by Kimmelman’s international stem-cell society, Fu’s team destroys the cells just five days after they’re made. This prevents the structures from developing what bioethicists term “features of concern”—such as a primitive nervous system.
But scientists are prepared to argue that their structures aren’t real embryos, and that they should be able to stretch the limit. Some experts are calling for an end to the rule altogether, saying it is outdated. John Aach, a scientist at Harvard Medical School, thinks entirely new ethical measuring sticks will be needed to help guide tests of organoids. For instance, could a mini-brain grown in the lab somehow feel suffering? And can our definition of an embryo withstand evidence that labs can make new sorts never before seen? “All great scientific advances have a way of exposing the imprecision of common concepts and forcing people to rethink them,” says Aach.
Even before his paper came out, Shao was buttonholing ethics experts, including Insoo Hyun, a professor at Case Western University, at a conference this year in Boston. Hyun felt the young researcher was on safe ground because his structure didn’t contain every part of an embryo. “I think that they should design experiments to focus on specific questions, and not model everything,” says Hyun. “My proposal is, just don’t make the whole thing. One team can make the engine, another the wheels. The less ambiguous morally the thing is that you are making, the more likely you can do your research unimpeded.”
There’s yet another reason to be cautious. The U.S. currently bars federal funding for any study of embryos, no matter how they are made, under a law called the Dickey-Wicker Amendment.
While today’s embryoids don’t appear to be covered by the legal restriction, they might be if scientists make them realistic enough. In response to written questions, the science policy office of the National Institutes of Health, the $33-billion-a-year funding agency, says it has an internal process it uses to analyze grants and to determine if “proposed research would create an organism that meets the statutory definition of a human embryo.”
The Michigan scientists, whose project used funds from two NIH grants, say agency officials haven’t raised any objections so far. For now, the embryoids live and die in boxes made of lucite and metal and are fed with culture medium. “Because of the really heavy engineering component to these entities, I think you will be able to argue these are not organisms,” says Hyun. That’s a point that Shao has sought to emphasize, too. When Shao presented the group’s work this year, he added to his slides an ethics statement outlined in a bright yellow box, saying the embryoids “do not have human organismal form or potential.”
But such definitions could be a moving target. The whole point of the structures is the surprising, self-directed, even organismal way they develop. Robert Cork is the head of the Virtual Embryo Project, which maintains the images the Michigan team used to identify their structures. When I asked him about Shao’s paper, Cork told me that the embryoids could go on to make some of the parts they’re currently missing, if the experiments were allowed to progress. “This would suggest that if they can keep the cysts viable for longer they might go ahead and start to develop into something more ‘embryo-like,’” says Cork.
High-throughput

Fu says the next step in his Ann Arbor laboratory is to perfect procedures for making embryoids with specific characteristics, and in larger numbers. Initially, of every 100 “cysts” the Michigan scientists grew, only five ended up with the asymmetric shape reminiscent of the amniotic sac. But they have already learned how to make that shape emerge every time. The production of embryoids will become “programmable and scalable,” Fu predicts.
Drugs could be tested on the embryoids, for instance to flag any that have toxic effects and cause birth defects. Fu’s hope is that synthetic embryology might eventually help engineers grow complete human organs. “I am not talking about a human body without a brain. But what is a true possibility is you could develop a mini-gut or a mini-liver, since the embryo develops them, too. And if you have the primitive organs, they could grow into a functional one,” Fu predicts. The lab has started growing embryoids on a chip about the size of a credit card. Etched into it are six microchannels, each accommodating 10 of the entities, which are suspended in hydrogels and fed nutrients held in miniature buckets. Fu calls it “high-throughput manufacturing.”
This way, he says, “everything can be triggered and under control.”
