Assistant Professor Allen Liu was featured in an article in The Scientist titled “The Mechanobiology Garage: New tools for investigating how physical forces affect cells.” In the article, The Scientist reports on recently developed methods—from upgraded versions of conventional tools to newer micro- and nanotechnologies—in the proliferating tool chest of cellular mechanobiology research.
From The Scientist:
Micropipette Arrays
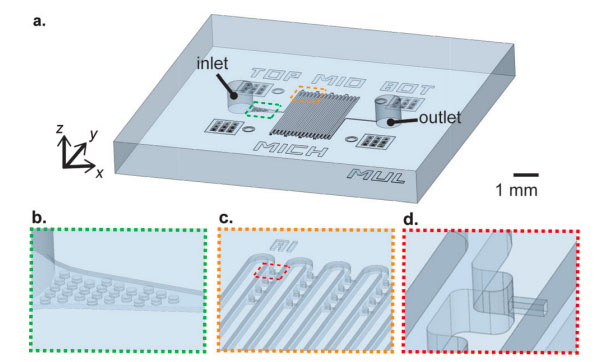
Problem: Micropipette aspiration of a section of an intact cell is a useful technique for measuring mechanical properties such as the cell’s stiffness, but setting up the experiment is time-consuming, and throughput is extremely low (~10 minutes/cell).
Solution: Liu and colleagues fabricated a microfluidic device consisting of a group of micropipettes that can perturb up to 128 cells simultaneously. They attached the micropipettes to a simple, cheap, calibrated pump that generates fluid pressure to exert forces on the cells through the micropipettes. The team then used two computational models to describe how the deformations produced by the device reflect mechanical properties of the cells. The researchers showed that, consistent with previous findings, the stiffness of cultured metastatic breast cancer cells was lower than that of normal breast epithelial cells (Lab on a Chip, 15:264-73, 2015).
Pros:
- The device is much cheaper (~$3,000) than a conventional micropipette aspirator machine (>$10,000) as it does not require expensive electronics such as a piezo motor, an electrical motor that senses deflections and is used for force calibration.
- It offers higher throughput than a conventional micropipette aspirator.
Cons:
- Without the electronics of the traditional micropipette, the tool cannot exert small-magnitude forces of under a few hundred nano-Newtons and has limited sensitivity.
- The micropipettes are grouped together and cannot be controlled individually. Channels can get clogged by debris, which can affect the measurements and system throughput.
- The device cannot be applied to mechanoreceptors on cellular extensions such as neurites of neurons, because the cells, which are detached during mechanical phenotyping, lose their extensions.
Future plans: Liu plans to develop a device to monitor mechanical cues in addition to pressure, to investigate how “multiple physical cues can orchestrate convergent mechanotransduction,” he says.
